Sodium channel gene takes diverging paths in autism, epilepsy
Mutations in a gene called SCN2A have opposite effects in autism and in epilepsy.
Mutations in a gene called SCN2A have opposite effects in autism and in epilepsy. The divergence makes the gene an attractive candidate for research, suggest unpublished results presented today at the 2016 Society for Neuroscience annual meeting in San Diego.
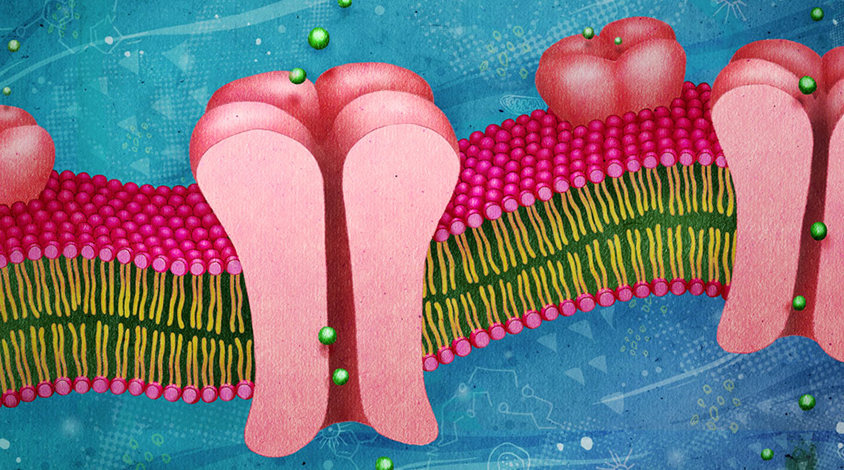
SCN2A has a single function: It encodes a channel that transmits sodium ions across a cell membrane. The channel sits along neuronal fibers, enabling the cells’ electrical signaling.
By contrast, many other autism candidates, such as CHD8, control thousands of other genes.
“We have known what sodium channels do since the 1950s,” says Kevin Bender, assistant professor of neurology at the University of California, San Francisco, who presented the findings.
“We have a wealth of basic neuroscience that we can build on.”
Mutations in SCN2A have long been known to cause severe forms of childhood epilepsy. The new work shows that the mutations found in people with autism are distinct from those seen in people with epilepsy.
The researchers first examined the effects of 25 mutations in SCN2A seen in people with autism. Most of these mutations in the gene change a single amino acid, which makes it difficult to infer their effect on the corresponding protein’s function. The researchers found that the mutations cluster either in a segment of the protein that sits inside the cell or in one that restricts the channel to sodium ions.
Different hits:
A literature search revealed that the SCN2A mutations linked to epilepsy are found in other sites: They are typically located in a part of the protein that sits within the cell membrane. This region senses when the channel should open.
Bender’s team also expressed 11 of the autism-linked mutations in cultured human kidney cells to measure how well the channels in those cells transmit ions. Eight of the mutations stop the channel’s activity; the remaining three diminish its ability to respond to signals.
By contrast, SCN2A mutations linked to epilepsy make the channels hyperactive. The findings make sense, as hyperactive neurons are thought to underlie seizures, Bender says.
The data suggest that the kinds of mutations found in epilepsy and autism differ not just in location but in their function. In support of this theory, preliminary data show that people with autism who have SCN2A mutations do not tend to have seizures.
In the case of autism, SCN2A mutations may affect development, Bender says. The gene is primarily expressed before 2 years of age. It is also located mainly in neurons that excite brain activity. SCN2A mutations may set up a signaling imbalance early in development that has long-term consequences.
For more reports from the 2016 Society for Neuroscience annual meeting, please click here.
Recommended reading
New tool may help untangle downstream effects of autism-linked genes
NIH neurodevelopmental assessment system now available as iPad app
Molecular changes after MECP2 loss may drive Rett syndrome traits
Explore more from The Transmitter
Organoids and assembloids offer a new window into human brain

Who funds your basic neuroscience research? Help The Transmitter compile a list of funding sources
