Genetic changes that damage PTEN, a gene strongly linked to autism, cause a buildup of brain fluid by prompting neural stem cells to proliferate and block a channel that drains the ventricles, a new mouse study finds.
Loss of PTEN protein—a key regulator of cell division—also drives a surplus of inhibitory interneurons that impairs cortical function in mice. Blocking the signaling pathway underlying this proliferation decreases these brain changes in mice, the study shows.
“The findings position [cerebrospinal fluid] not merely as a passive cushioning medium but potentially as an active regulator of brain development,” says Hyun Kyoung Lee, associate professor of pediatrics and neurology at Baylor College of Medicine, who was not involved in the study.
The work adds to evidence of a link between autism and excess brain fluid. Younger siblings of children with autism—who are 20-fold more likely to be diagnosed themselves than are children in the general population—have increased cerebrospinal fluid, previous research has shown.
And 1 in 5 children with congenital hydrocephalus—enlarged ventricles containing excess brain fluid—also has autism. The condition is treated by surgically diverting fluid away from the brain. The procedure—known as shunting—reduces ventricular volume but does not ease autism traits.
The new findings explain this strategy’s ineffectiveness, at least in cases that involve PTEN variants: Although the surgery restores typical levels of cerebrospinal fluid, PTEN’s absence still skews neuronal numbers in the cortex and impairs cortical function, says Bryan Luikart, professor of neurobiology at the University of Alabama and one of the study’s principal investigators.
V
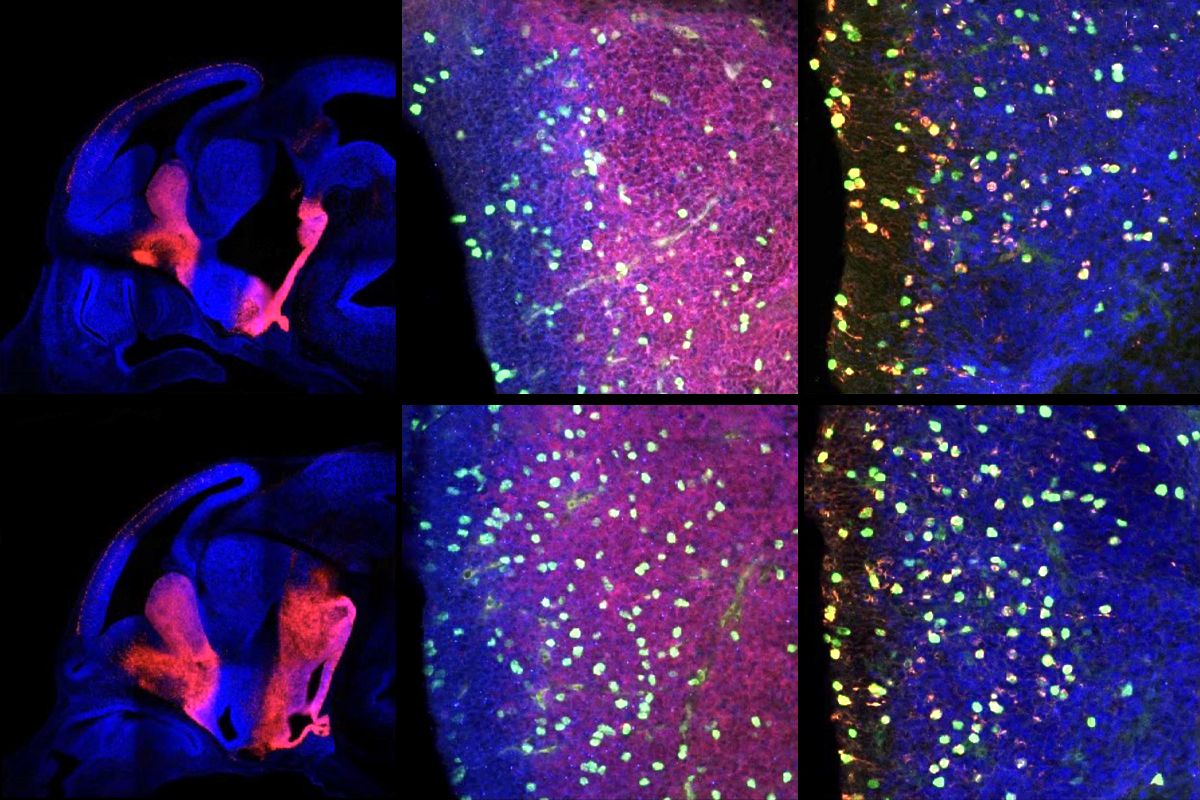
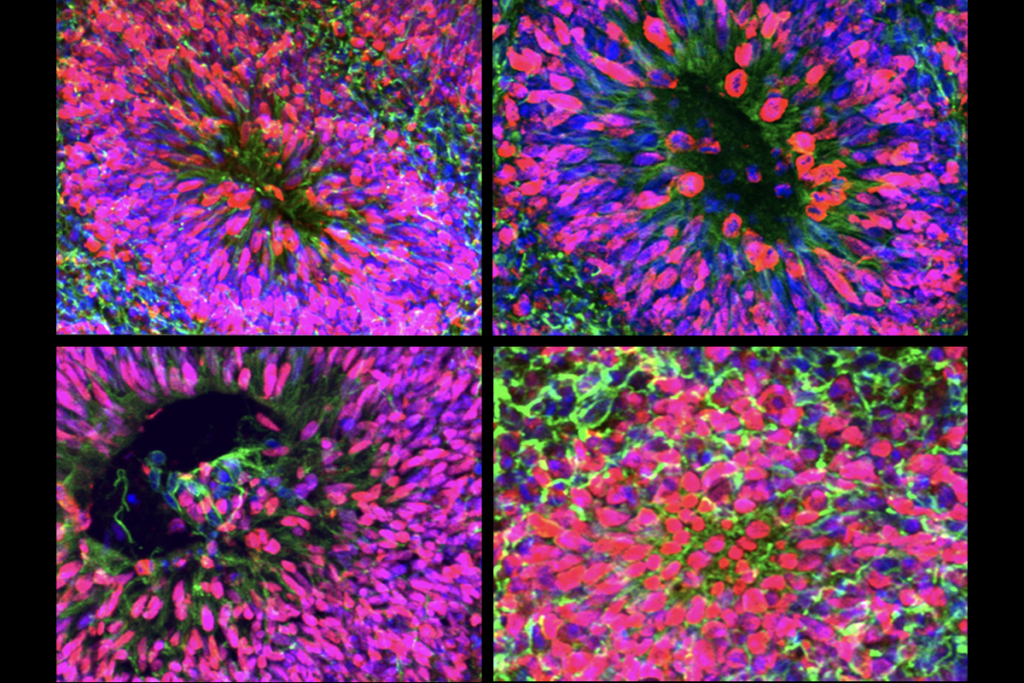
ariants in five genes, including PTEN, mTOR and PI3KA—components of the PI3 kinase signaling pathway—are more common than would be expected by chance among the whole-exome sequences of 482 people with enlarged ventricles who previously underwent shunting, the study reveals. PTEN emerged as the top altered gene in a subsequent analysis of genetic data from 2,697 children with congenital hydrocephalus.The team engineered mice missing PTEN in neural precursor cells in the medial ganglionic eminence, which give rise to cells on the ventricles’ periphery and inhibitory interneurons in the surrounding cortex. One day after birth, these mice had enlarged ventricles containing excess fluid compared with their wildtype littermates, MRI scans showed.
That swelling is caused by cell hyperproliferation and blockage of the channel that siphons away brain fluid, further imaging experiments revealed. And the animals’ choroid plexus—the network of cells and vessels that line the ventricles—exuded three times as much fluid as wildtype mice, the study found.
Mice lacking PTEN have more parvalbumin-expressing interneurons, a type of neuron that helps to pattern brain activity, the study also found. When Luikart and his colleagues tickled the animals’ whiskers during calcium imaging, they found hyperconnectivity in a part of the somatosensory cortex, and underconnectivity in a motor region. The findings hint that altered activity of parvalbumin-positive interneurons contributes to sensory traits by influencing cortical connections, Luikart says.
Deleting a component of mTORC1, a protein complex known to be hyperactive in people with PTEN variants, or treating the animals with the mTORC1 inhibitor everolimus reduces ventricular swelling, the study found.
The drug, which is used as an immunosuppressant and cancer therapy, could offer a nonsurgical alternative for treating children with excess brain fluid, says Ying Ni, assistant professor of medicine at Case Western Reserve University, who was not involved in the study.
The findings were published on 24 February in Nature Neuroscience.
But mTORC1 is “probably not the whole story,” Luikart says. Everolimus treatment among people with PTEN-linked autism yielded few improvements in an independent team’s 2022 phase II clinical trial. The results might reflect some of the participants’ advanced age—the treatment appears to be better at preventing rather than reversing brain changes—and the fact that some of PTEN’s effects are mediated by the related complex mTORC2, he says.
Some of the study participants have PTEN-linked hydrocephalus without autism, but it’s unclear why, Luikart says. One possibility is that different variants contribute to ventricular changes or cortical changes to different extents—something Luikart says he and his collaborators hope to explore further by comparing variant-specific mouse models.
The team also aims to understand the interaction between cerebrospinal fluid and cortical function in animals with PTEN variants, Luikart says. Initial evidence suggests the two are intertwined: Synchronized neuronal activity generates rhythmic waves that help brain fluid flow and wash away metabolic waste, according to a 2024 paper.