Linking autism, sex, gender and prenatal hormones
Elevated levels of fetal sex steroid hormones such as testosterone may explain many of autism’s unique features.
Why do so many people prefer the term ‘gender’ to ‘sex’? Gender refers to your identity, to how masculine or feminine you feel yourself to be. Gender seems plastic; some would even say it’s a matter of choice. By contrast, sex seems more deterministic: It is defined by your chromosomal makeup, whether you have one X and one Y as males do, or two X’s as females do.
Gender carries with it an optimistic connotation, that anyone can be anything they want and write their own destiny — so it’s not surprising that many people prefer to use it over sex. Because biological and sociocultural factors are inextricably linked, however, scientists are increasingly using the term ‘sex/gender.’
Social experience and genetics are not the only two factors that determine our sex/gender. A third, increasingly important, factor is the contribution of prenatal sex steroid hormones, such as testosterone. There is growing evidence that elevated prenatal testosterone may lead to the raft of differences seen in people with autism when compared with typical individuals.
We’ve known about the importance of testosterone in ‘masculinizing’ not only the reproductive system but also the brain and behavior for almost 70 years. It’s worth reviewing the classic early experiments. In 1947, endocrinologist Alfred Jost castrated a male rabbit fetus, depriving him of his testosterone, and discovered that the rabbit then developed a female-typical reproductive system — fallopian tubes, a uterus and a vagina1.
In 1953, Jost did the reverse experiment2. He discovered that implanting testes into a female rabbit fetus — which increases her testosterone exposure — causes the rabbit to develop a male-typical reproductive system. In 1959, Charles Phoenix’s now classic experiments in guinea pigs found that castration causes a male guinea pig to show female-typical mating behavior, and giving extra testosterone causes a female to show male-typical mating behavior3.
Male brain:
We’ve known since 1966 that an oval-shaped region of the hypothalamus called the sexually dimorphic nucleus controls mating behavior, and that this region is larger in males than in females. If you give a female rat extra testosterone at birth, this region in her brain grows as large as that of a typical male.
Testosterone doesn’t work alone in ‘masculinizing’ the brain, however. For example, testosterone is aromatized into estrogen, and in 1976 Dominique Toran-Allerand and her colleagues showed that exposure to excess estradiol at birth increases the number of neurons, and the connections among them, in a female rat’s brain4.
What does this have to do with autism? We know that the brains of people with autism have a high number of dendritic spines — the neuronal protrusions that receive messages — because of decreased pruning in the brain. We also know that elevated fetal testosterone reduces pruning. But is there evidence of elevated fetal testosterone in autism?
Earlier this year, my colleagues and I tested this by looking at hormone levels in samples of amniotic fluid from 128 boys later diagnosed with autism and 217 controls. We found that, indeed, samples from the boys with autism show elevated levels of testosterone, along with other hormones that are part of the so-called delta-4 sex steroid pathway5.
If testosterone and other androgens are elevated prenatally in autism, we should expect to see a raft of consequences for their physiology, brain and behavior. Published studies have confirmed the growing list of consequences outlined below6.
Clear consequences:
People with autism should show higher rates of delayed language, reduced eye contact and lower empathy because all of these are inversely correlated with prenatal testosterone levels. They should show superior attention to detail and a stronger interest in systems because both of these are positively correlated with prenatal testosterone.
In women with autism, we should see higher rates of polycystic ovary syndrome — characterized by delayed puberty, irregular menstrual cycles and hirsutism (excess bodily hair) — because the syndrome is also associated with higher levels of prenatal testosterone7. We should also see gender variance and higher rates of tomboyism8. And we should see masculinized 2D:4D (the ratio of the second to the fourth digit length) measures, because this correlates with high prenatal testosterone.
Women with autism should have a ‘masculinized’ brain in terms of gray and white matter, and people with autism overall should show an extreme of the male pattern of scores on psychological tests of empathy and systemizing, because each of these are associated with high levels of prenatal testosterone9. Finally, we should see elevated levels of testosterone or its precursors10.
Testosterone is a sex steroid hormone, so this theory of autism is called the prenatal sex steroid theory. It gains further support from an animal model in which giving rats a drug called letrazole — which elevates testosterone levels — causes rats to socialize and vocalize less11. The fact that prenatal testosterone is epigenetic — meaning that it can alter gene expression without altering the genetic code — makes its role even more important, providing a new window into gene-hormone interactions.
Simon Baron-Cohen is director of the Autism Research Centre at Cambridge University in the U.K.
References:
- Jost A. Exp. Biol. Med. 66, 302-303 (1947) Abstract
- Jost A. Recent Prog. Horm. Res. 8, 379-418 (1953)
- Phoenix C.H. et al. Endocrinology 65, 369–382 (1959) PubMed
- Toran-Allerand C.D. Brain Res. 106, 407-412 (1976) PubMed
- Baron-Cohen S. et al. Mol. Psychiatry 20, 369-376 (2015) PubMed
- Baron-Cohen S. et al. PLoS Biol. 9, e1001081 (2011) PubMed
- Pohl A. et al. Mol. Autism 5, 27 (2014) PubMed
- Strang J.F. et al. Arch. Sex. Behav. 43, 1525-1533 (2014) PubMed
- Baron-Cohen S. et al. PLoS One 9, e102251 (2014) PubMed
- Schwarz E. et al. Mol. Psychiatry 16, 1213-1220 (2011) PubMed
- Xu X.J. et al. Physiol. Behav. 138, 13-20 (2015) PubMed
Recommended reading
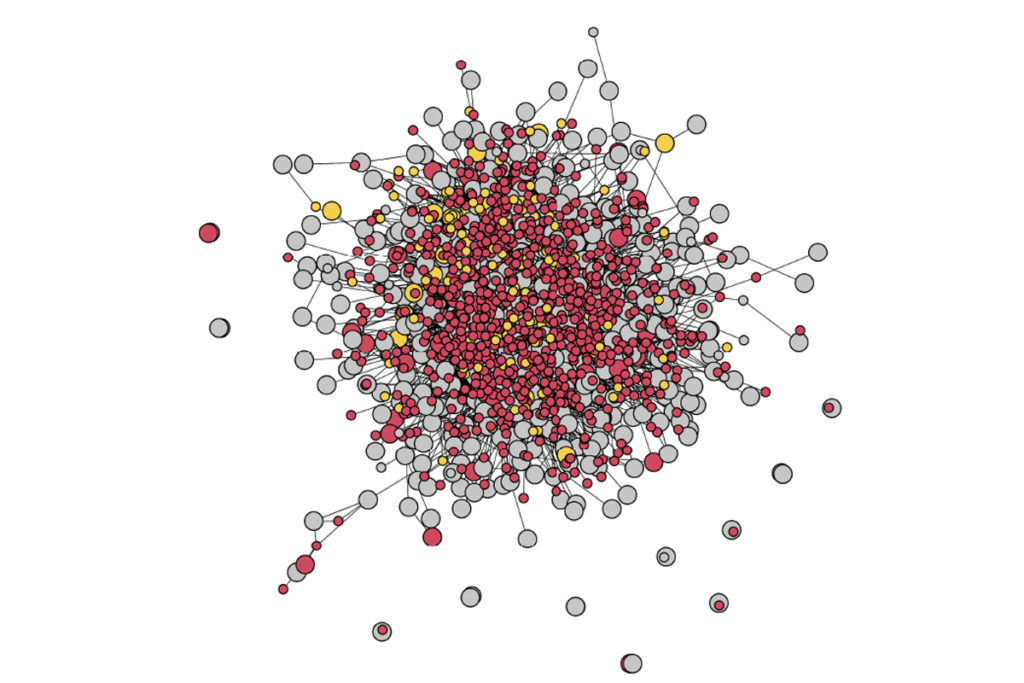
Sequencing study spotlights tight web of genes tied to autism
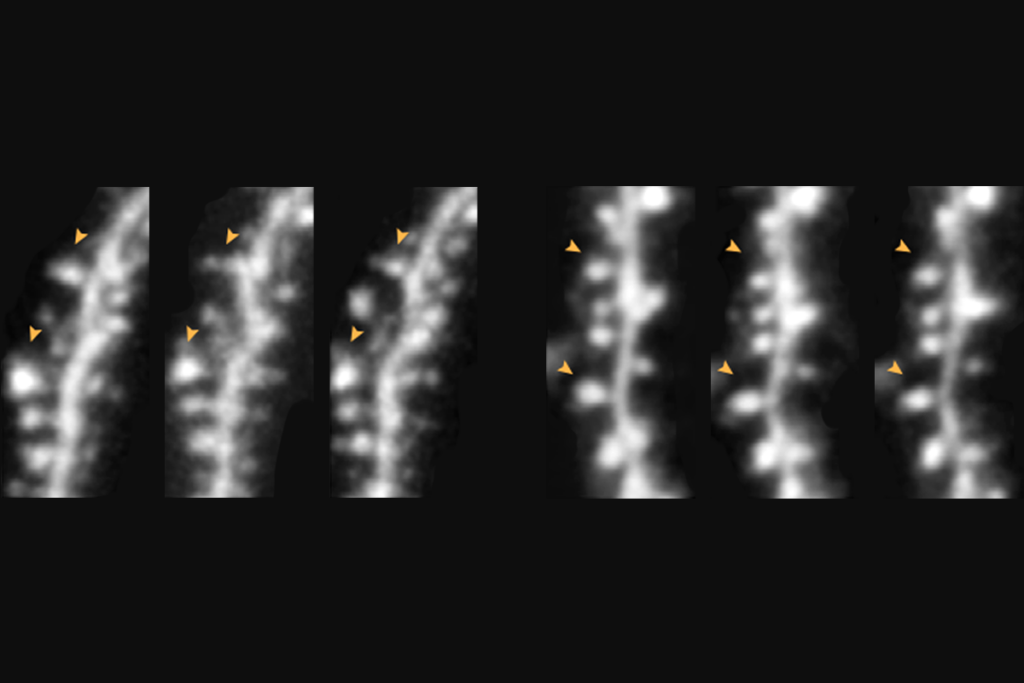
Targeting NMDA receptor subunit reverses fragile X traits in mice

Maternal infection’s link to autism may be a mirage
Explore more from The Transmitter
Alison Preston explains how our brains form mental frameworks for interpreting the world

Calculating neuroscience’s carbon cost: Q&A with Stefan Pulver and William Smith
