Repeat scans reveal brain changes that precede childbirth
A detailed look at a “pregnant brain” highlights a need to investigate the neural alterations that occur during a transition experienced by nearly 140 million people worldwide each year.
When neuroscientist Liz Chrastil decided to try to get pregnant using in vitro fertilization in 2019, she came up with an idea for a new study: to scan her brain before, during and after pregnancy.
Such detailed scans had never been done before. A few neuroimaging studies had suggested that people show cortical volume decreases during pregnancy, but those studies had primarily scanned the participants before conception, once (if at all) during pregnancy and again after they had given birth, leaving the gestational period itself largely overlooked.
Chrastil says she was inspired by a 2020 study in which researchers had scanned the brain of one woman every day for 30 days, revealing how functional connectivity changes across the menstrual cycle. So she contacted that study’s lead investigator, Emily Jacobs, to see if she and her lab would like to try the same type of dense sampling on a pregnant person.
“Everyone was on board immediately,” says Chrastil, associate professor of neurobiology and behavior at the University of California, Irvine.
Chrastil underwent 26 MRI scans in total—four pre-pregnancy, at least four during each trimester and another seven during the two years after she gave birth—along with 19 blood draws to assess her sex hormone levels.
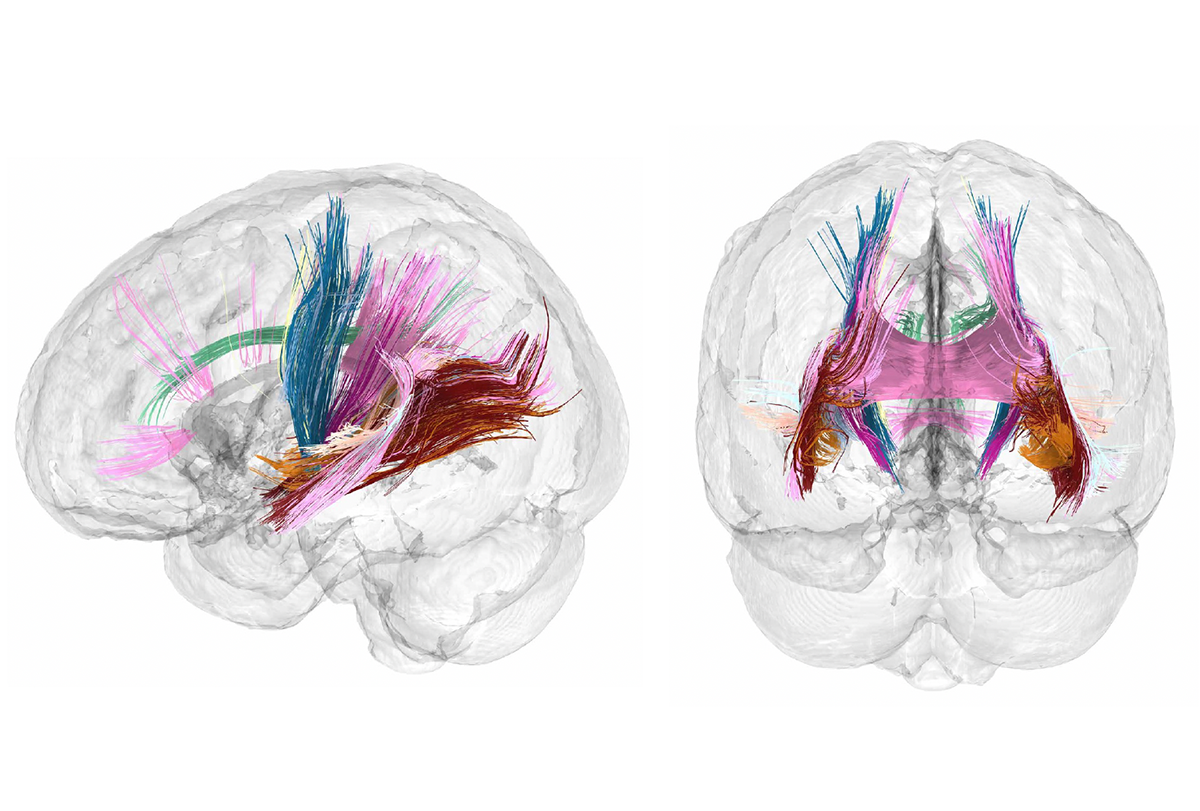
The scans confirm the shrinkages reported in previous studies and reveal for the first time how those changes unfold from week to week across the brain and in step with shifts in estradiol and progesterone levels, Chrastil and her colleagues report in a paper out today in Nature Neuroscience. The study also shows a transient increase in the structural integrity of white-matter tracts during the first and second trimester, which returns to baseline after childbirth.
It is clear there is neuroplasticity during pregnancy, says Jacobs, director of the Ann S. Bowers Women’s Brain Health Initiative at the University of California, Santa Barbara. “And now the question is: Why are these changes happening?”
P
art of the reason this gestational “extended neuroplasticity” is being revealed only now, Jacobs says, is because pregnant women rarely have their brain scanned. “I think there’s this fear that we can’t scan pregnant women,” she says, even though peer-reviewed studies have found that MRI poses no risks to a fetus.The first hint that pregnancy warranted a closer look came in 2016, when a team of scientists in Europe scanned 25 first-time mothers during the year before, and again two years after, pregnancy. In brain regions linked to social cognition, gray-matter volume was lower in these women than in 20 controls, the team found. These changes were still visible six years postpartum, according to a 2021 follow-up study of seven of the mothers and five controls.
Cortical volume decreases are present before childbirth, researchers reported in a January study. Although decreases in cortical thickness attenuate postpartum, changes in certain areas of the brain, such as those that contribute to the default mode network, may never revert to the pre-pregnancy state, says Susana Carmona, a neuroscientist at Gregorio Marañón Health Research Institute in Madrid, who worked on the previous studies but not Chrastil’s.
The decreases in cortical gray-matter volume and thickness across Chrastil’s brain occurred in 80 percent of the 400 measured regions. They appeared at the beginning of pregnancy, steadily shrank “in step” with gestational week and were nearly three times greater than the changes seen in eight controls scanned over the same time period. A recent preprint that includes within-pregnancy scans from Carmona’s lab shows similar results—a nearly 5 percent decrease in gray-matter volume during pregnancy.
Chrastil’s changes in gray-matter volume and cortical thickness recovered slightly after childbirth but persisted two years postpartum. Other metrics, such as the volume of her lateral ventricles and that of her cerebrospinal fluid, increased in a nonlinear fashion, rising in the second and third trimesters and dropping “sharply” postpartum, according to the study.
What was more surprising, Chrastil says, “was the white matter, which showed this interesting nonlinearity.” Its structural integrity increased during the first and second trimesters but returned to baseline postpartum, the study shows. This trajectory did not appear in previous research that collected only pre- and postpartum scans.
“I’m a big fan of the approach,” says Nico Dosenbach, associate professor of neurology at Washington University School of Medicine in St. Louis, who was not involved in Chrastil’s study. Because the study was done on one person, the strong correlations between brain measures and week of gestation are “a whole different level of believability, because you don’t have to worry about registration errors”—which can occur when brain images are aligned across multiple people for analysis.
T
he next line of inquiry involves what these changes mean from a functional perspective, Jacobs says—for instance, “Can we use this precision imaging lens to understand somebody’s risk for postpartum depression or to understand the impact of preeclampsia on the brain?”Figuring out the cellular and molecular mechanisms causing these brain changes in pregnancy is a vital part of the future of this work, too, says Magdalena Martínez-García, a postdoctoral researcher in Emily Jacobs’ lab at the University of California, Santa Barbara, who was not involved in Chrastil’s study.
Hormones are likely one piece of the puzzle, as they “are these small, innocuous molecules, yet they can completely change circuit functional behavior,” says Jonny Kohl, a group leader at the Francis Crick Institute, who was not involved in this study. Estradiol and progesterone prepare the female mouse brain for parenting because they act on neurons in the medial preoptic area, which is critical for parental behavior, according to a 2023 paper from Kohl’s lab published in Science.
In keeping, Chrastil’s cortical gray-matter volume and thickness changes occurred as the levels of estradiol and progesterone increased throughout pregnancy.
A greater appreciation “of the power of steroid hormones for shaping the brain is probably going to be a good thing,” Jacobs says. A next step in this research should involve “a finer-grain lens to understand really the biological drivers” of changes in the brain, she says.
Although hormones likely play a big role in the neuroplasticity in pregnancy, many other “life events” happen at the same time as becoming a parent, says Amy Kuceyeski, professor of mathematics in radiology at Weill Cornell Medicine, who was not involved in this study. She says she hopes new imaging studies include other groups, such as adoptive parents, to disentangle the effects of stress and other factors from that of pregnancy hormones.
Overall, Carmona and others say, the field needs more quality scan data to understand neuroplasticity during pregnancy, both causes and effects. One of the goals of assembling a new longitudinal cohort in her 2024 study, Carmona says, is to start with a larger sample size, which will hopefully make it easier to have a sizable group of participants to track long term, to better understand which of these brain network changes are permanent.
Chrastil and Jacobs are recruiting more women to participate in a detailed study of brain changes during pregnancy, in which they plan to draw blood and conduct MRI scans dozens of times, from before pregnancy until one year postpartum.
C
hanges in the “mothering brain” have broad implications for many other areas that neuroscientists are interested in studying, says Darby Saxbe, professor of psychology at the University of Southern California, who was not involved in these studies. “Social cognition, attention, close relationships, mental health, sleep—all of these things kind of intersect with changes that occur over the transition to parenthood.”The findings are prompting some researchers who use neuroimaging to rethink how they view some of their datasets and analyses, Saxbe says. “We screen out for handedness, but we don’t always look at parenting or reproductive history,” in neuroimaging studies.
For now, publicly available neuroimaging datasets “are not focused on collecting women’s brain health and reproductive health information,” Kuceyeski says. Large, open neuroimaging datasets are adding measures of blood hormones to datasets, so improvements are “in the pipeline,” she adds.
Martínez-García agrees: “I think another important thing to say to every neuroscientist is, if you’re working with humans, and they are adults, please collect this data.”
The current research on the neuroscience of pregnancy reminds Jacobs of similar work that revealed the neuroplasticity of the adolescent brain. “I think pregnancy is going to be the same thing,” she says. “We’re going to realize that this is a really cool window in which the human brain has the capacity for some pretty striking remodeling.”
Explore more from The Transmitter

Exon-skipping approach boosts levels of key Rett syndrome protein